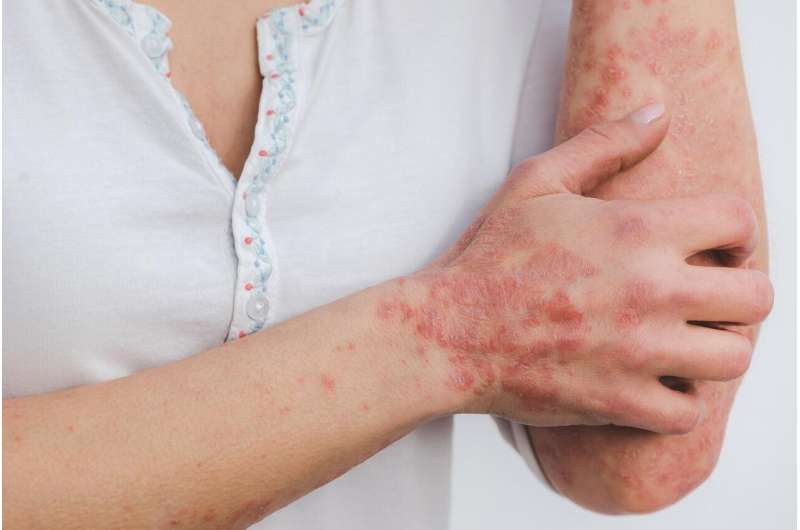
Birmingham scientists have made a significant breakthrough in the treatment of psoriasis, a chronic skin condition characterized by rapid skin cell proliferation, resulting in itchy and painful patches. Their study, published in Pharmacological Research, demonstrates that a sequence of just three amino acids can reduce the severity of psoriasis when applied topically in an emollient cream, potentially rivaling traditional steroid treatments.
Understanding Psoriasis
Psoriasis affects millions worldwide and presents a range of symptoms that can severely impact quality of life. The condition is driven by an overactive immune response and may lead to various physical and psychological issues, including:
- Raised scaly patches that are often red and inflamed.
- Itchiness and discomfort that can interfere with daily activities and sleep.
- Painful lesions that may crack, bleed, or ooze.
Typically, psoriasis is treated with various methods, including emollients and topical steroids, but these treatments come with risks of side effects, especially with prolonged use.
Introduction to PEPITEM
The focus of this research is on a molecule known as PEPITEM, which is a natural peptide in the body that regulates inflammation. This study narrowed down its functionality to a tripeptide sequence, which comprises three amino acids:
| Amino Acid Position | Amino Acid |
|---|---|
| 1 | Arginine (Arg) |
| 2 | Glutamic Acid (Glu) |
| 3 | Threonine (Thr) |
In their study, researchers led by Professor Ed Rainger from the University of Birmingham and Professor Francesco Maione from the University of Naples Federico II, discovered that this optimized tripeptide sequence and the full-length PEPITEM were effective in reducing inflammation in psoriasis models.
Comparing Efficacy
Through their experiments, the scientists observed that both the tripeptide sequence and the whole PEPITEM molecule resulted in a significant reduction in psoriasis severity, comparable to the leading steroid cream, Clobetasol Propionate 0.05%. To assess this, they utilized the PASI (Psoriasis Area and Severity Index), a standardized tool used in clinical practice to evaluate psoriasis severity:
| Treatment Type | PASI Score Reduction (%) |
|---|---|
| Tripeptide Sequence | 50% |
| Full-Length PEPITEM | 50% |
| Clobetasol Propionate | 50% |
Professor Ed Rainger emphasized the importance of developing new therapeutic agents that can be continuously used without the risk of side effects associated with steroid therapies. “Our findings raise the possibility of using PEPITEM-derived peptides for the treatment of psoriasis,” he noted.
Future Implications
The study opens up new avenues for psoriasis treatment, suggesting that PEPITEM-derived peptides may be used in tandem with other therapies. Such an approach could enable lower dosages of conventional treatments, potentially minimizing side effects through a steroid-sparing methodology.
Furthermore, the wider implications of the study suggest potential applications in treating other diseases characterized by chronic inflammation, such as rheumatoid arthritis, diabetes, and lupus.
“We have identified the parts of the PEPITEM molecule that are responsible for its biological action, and delivered peptides that mimic PEPITEM, dramatically influencing the skin's inflammatory processes.” – Professor Ed Rainger
Conclusion
The research conducted by the Birmingham team signifies a landmark step in the quest for safer and more effective treatments for psoriasis. The potential for these naturally derived peptides to mitigate the severity of this chronic condition without the side effects of long-term steroid use marks a promising development in dermatological therapeutics.
Reference
Saviano, A., et al. (2025). PEPITEM, its tripeptide pharmacophores and their peptidomimetic analogues regulate the inflammatory response through parenteral and topical dosing in models of peritonitis and psoriasis, Pharmacological Research. Retrieved from https://medicalxpress.com/news/2025-04-tiny-peptide-sequence-rivals-steroid.html
This document is subject to copyright. For more information regarding usage rights, please refer to the original publication.










Discussion